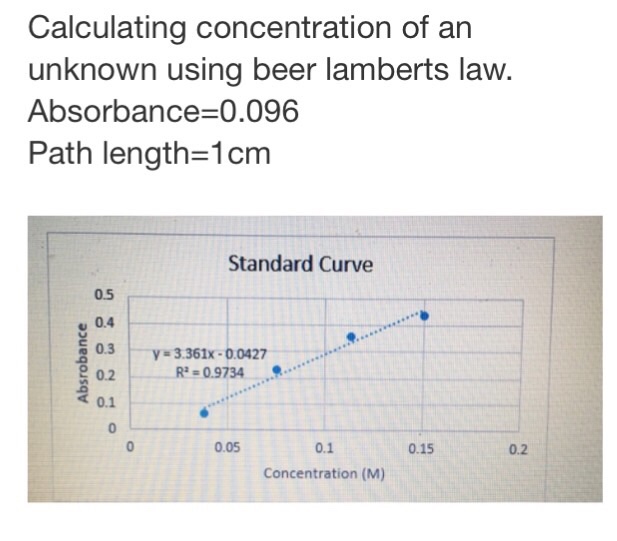
Worked example: Calculating concentration using the Beer–Lambert law | AP Chemistry | Khan Academy - YouTube

Beer Lambert's Law, Absorbance & Transmittance - Spectrophotometry, Basic Introduction - Chemistry - YouTube

Absorbance Measurements – the Quick Way to Determine Sample Concentration - Eppendorf Handling Solutions
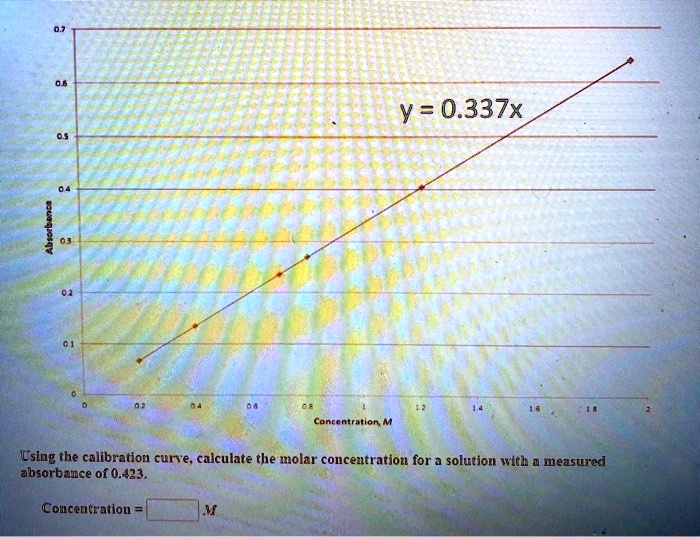
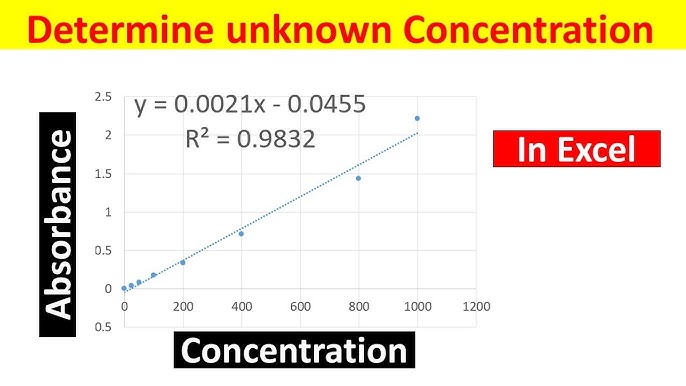
SOLVED: Y = 0.337x Conclusion: Using the calibration curve, calculate the molar concentration for a solution with a measured absorbance of 0.423. Concentration:

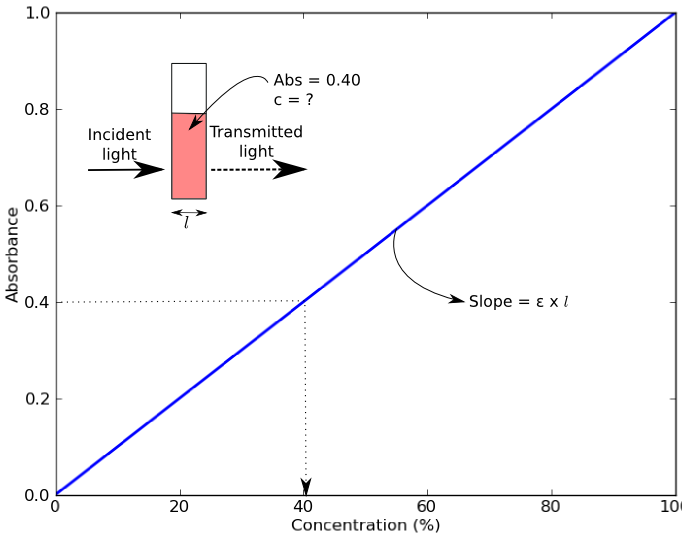
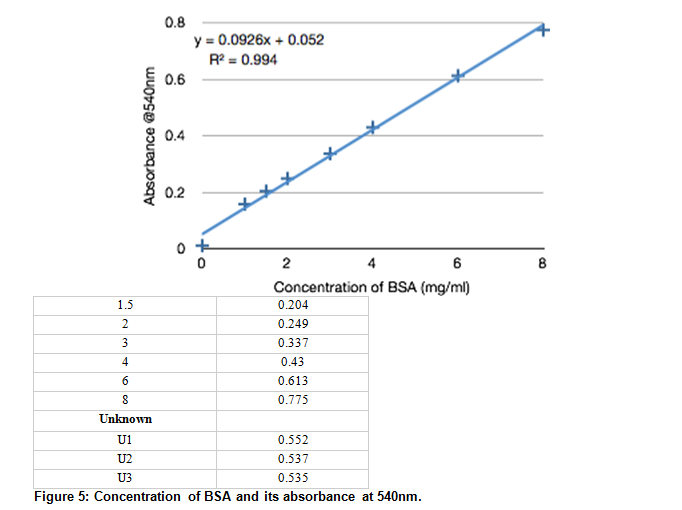
a) The absorbance of mixture at 400 nm was 0.40. Using the standard curve, calculate the concentration - brainly.com

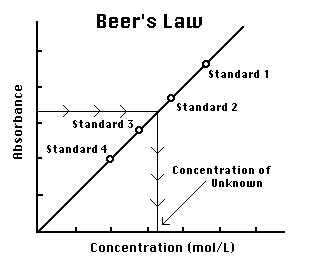
Use the data to plot a graph of Absorbance versus Concentration. Determine the concentration of the dye which would correspond to an absorbance of 0.140. What does Beer's Law say about the