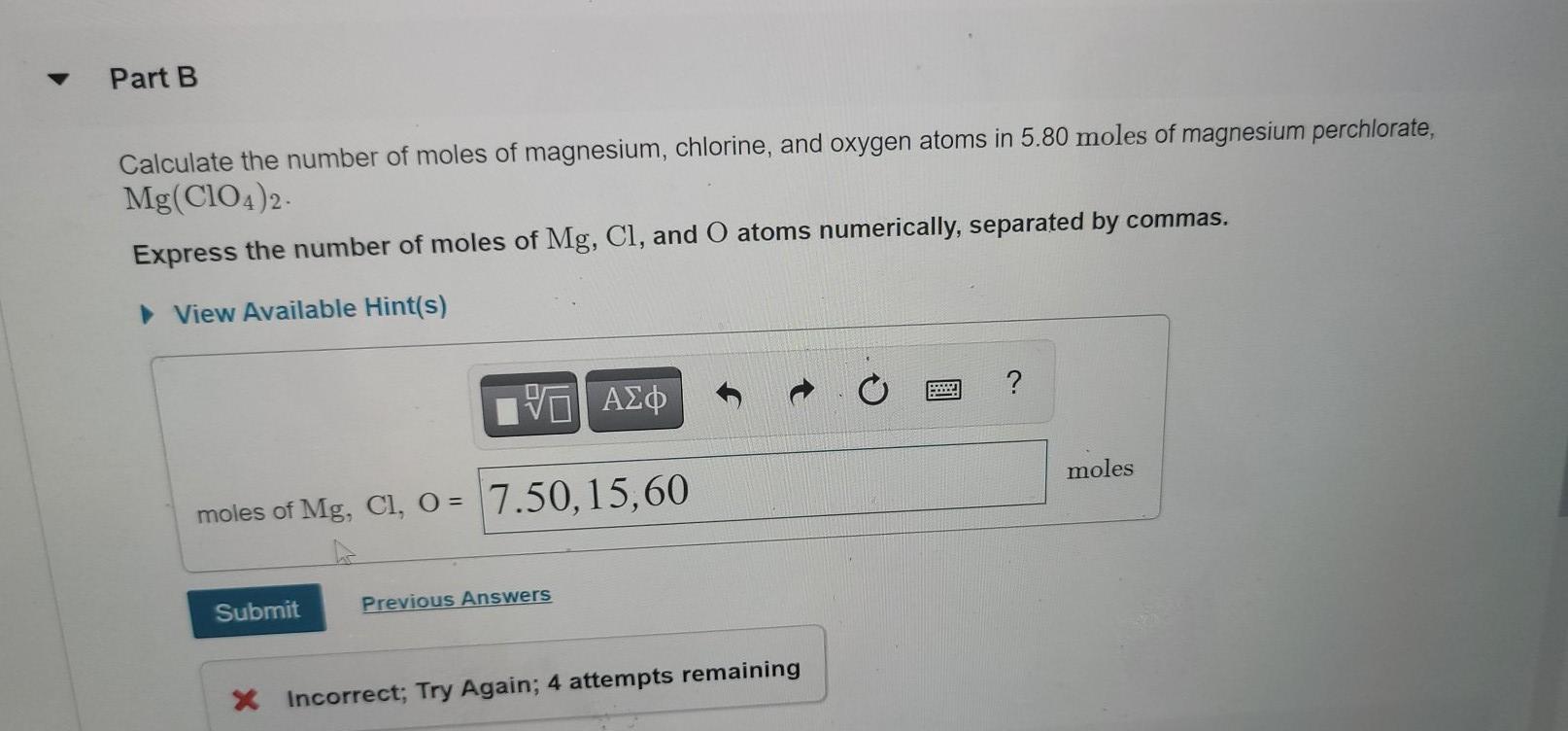
Calculate the mole ratio of `240 g` of calcium and `240 g` of magnesium. `(Ca = 40 , Mg = 24 u)` - YouTube
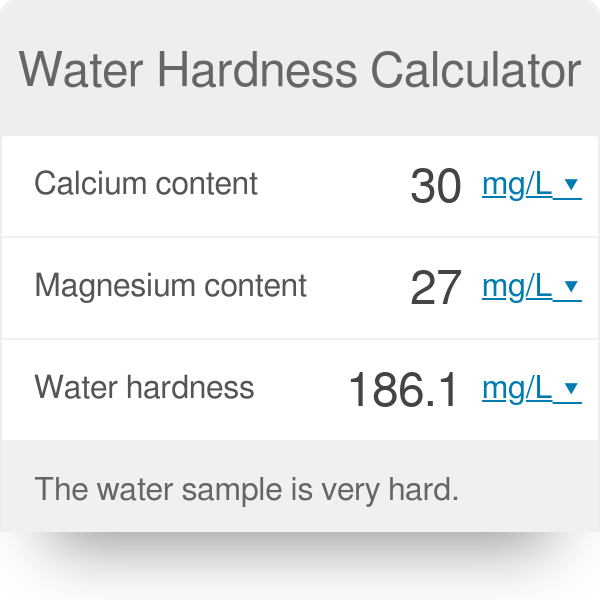
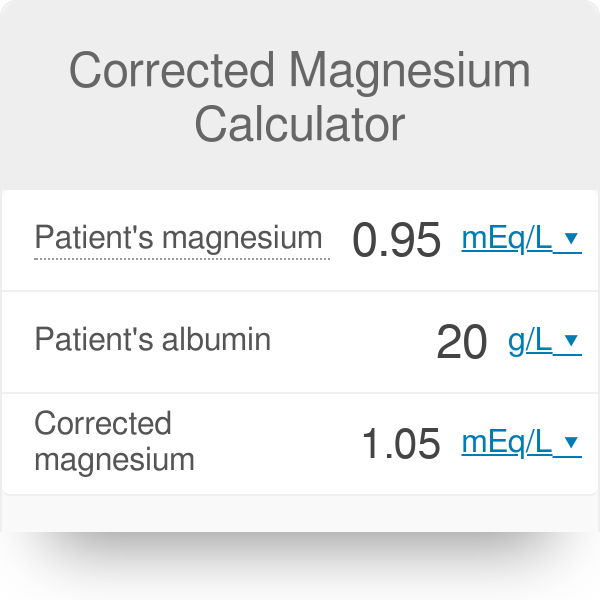
GitHub - Abhijeetbyte/MgCalculator: A web-based application specific calculator, used to calculate the amount of magnesium in water by evaluating other parameters.
![Calculate the number of molecules present in 0.5 moles of magnesium oxide (MgO) . [Atomic weights : Mg = 24, O = 16 ] Calculate the number of molecules present in 0.5 moles of magnesium oxide (MgO) . [Atomic weights : Mg = 24, O = 16 ]](https://toppr-doubts-media.s3.amazonaws.com/images/9335701/877be613-774e-48a4-af41-0d7e1c19f562.jpg)
Calculate the number of molecules present in 0.5 moles of magnesium oxide (MgO) . [Atomic weights : Mg = 24, O = 16 ]

Average atomic mass of magnesium is `24.31`amu. This magnesium is composed of 79 mole % of `24mg... - YouTube

View question - Calculate the mass of magnesium oxide formed when 0.52g of magnesium is burned according to the following equation: 2 Mg^s + O^2 ---> 2 MgO^s
![Calculate the number of molecules present in 0.5 moles of magnesium oxide (MgO) . [Atomic weights : Mg = 24, O = 16 ] Calculate the number of molecules present in 0.5 moles of magnesium oxide (MgO) . [Atomic weights : Mg = 24, O = 16 ]](https://dwes9vv9u0550.cloudfront.net/images/2527137/b4ebf9c0-6b64-43b4-94e9-555f3180cc58.jpg)
Calculate the number of molecules present in 0.5 moles of magnesium oxide (MgO) . [Atomic weights : Mg = 24, O = 16 ]