Novartis data show more patients are completely symptom-free from chronic spontaneous urticaria with ligelizumab (QGE031) than Xolair® 300 mg | Novartis

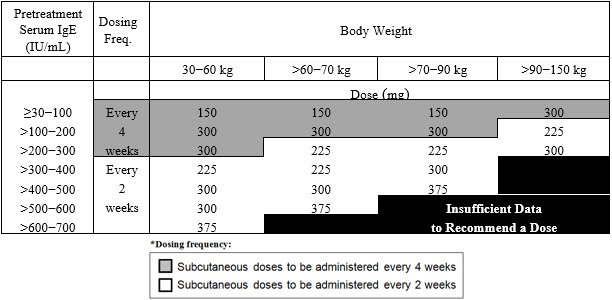
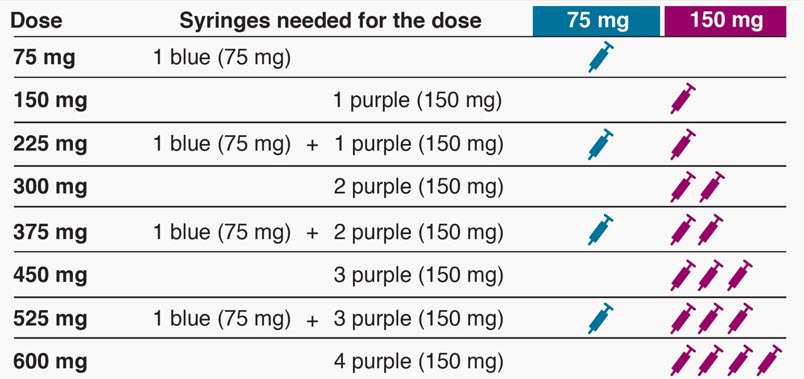
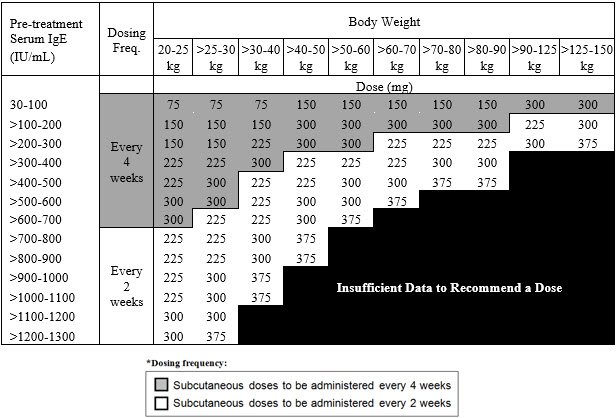
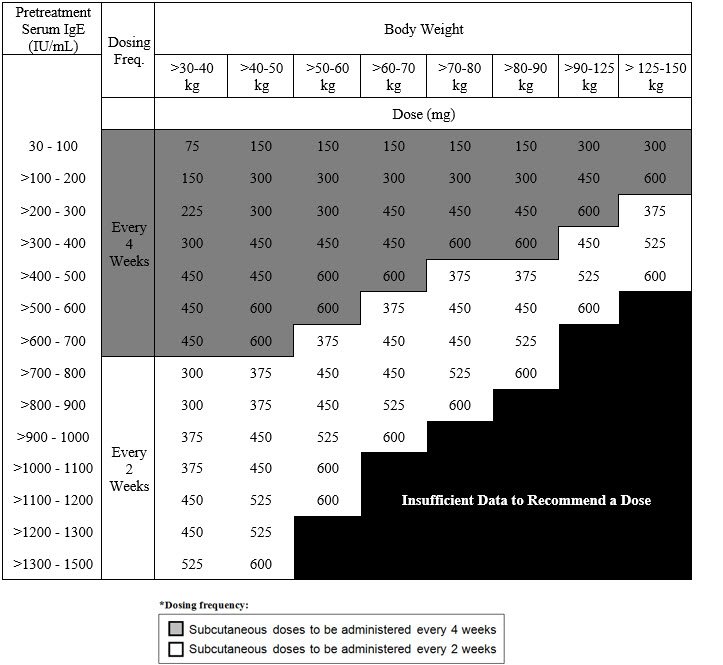
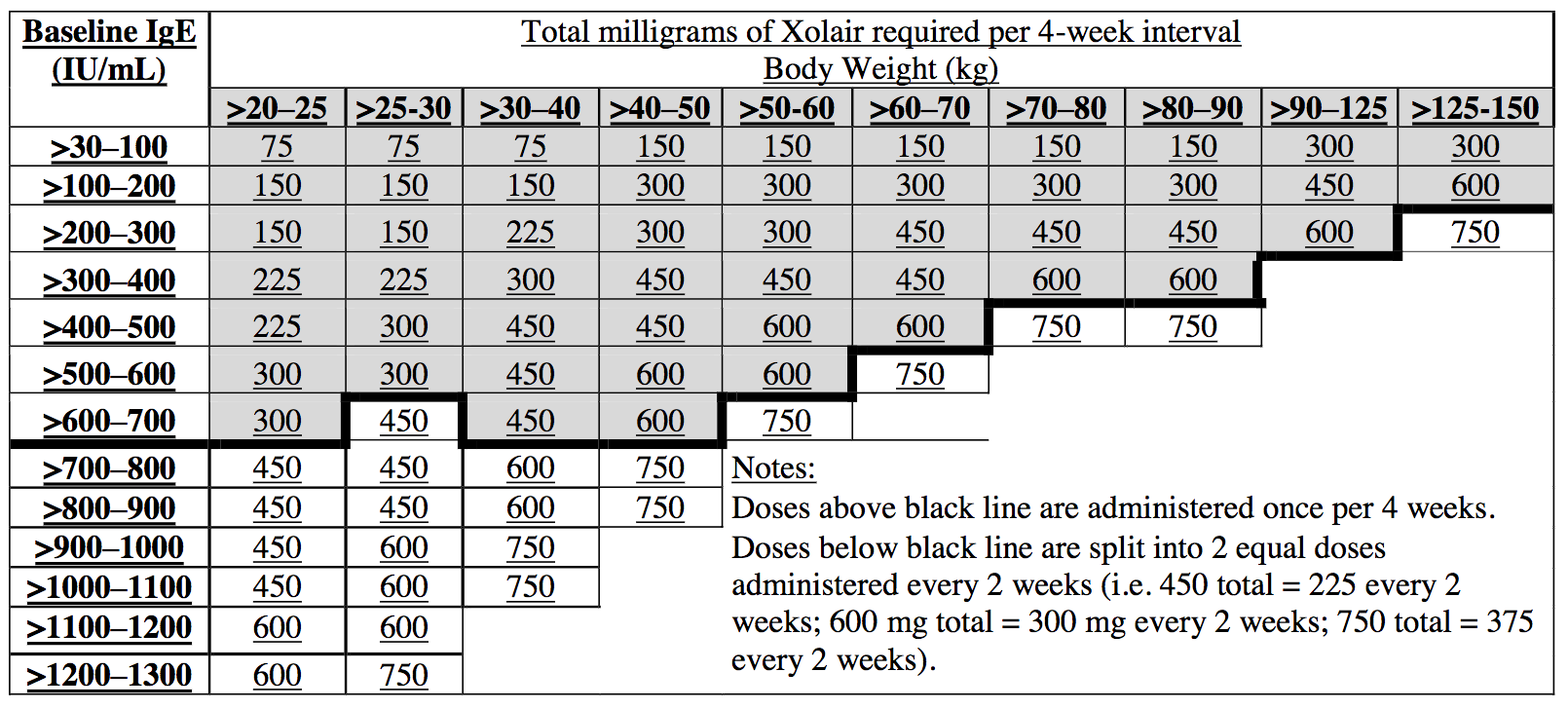
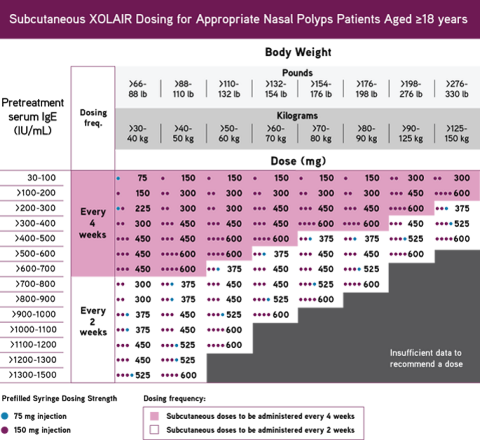
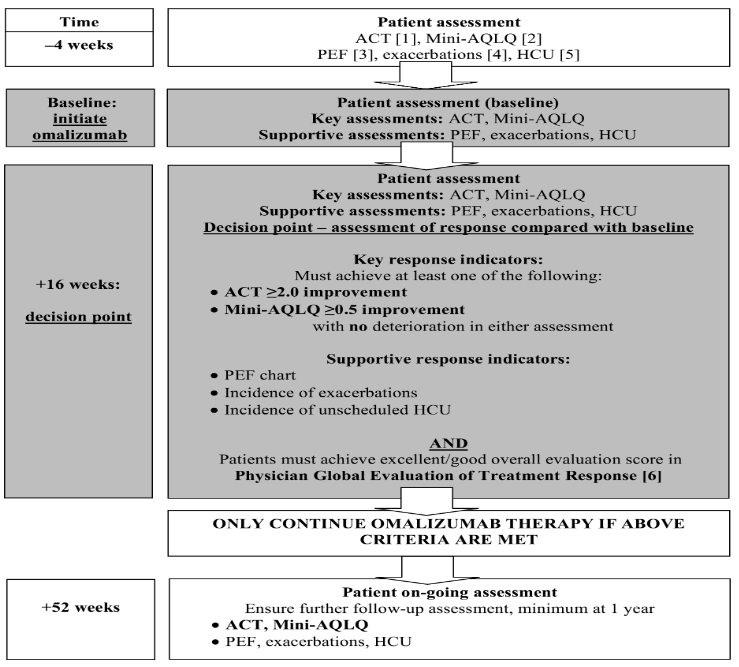
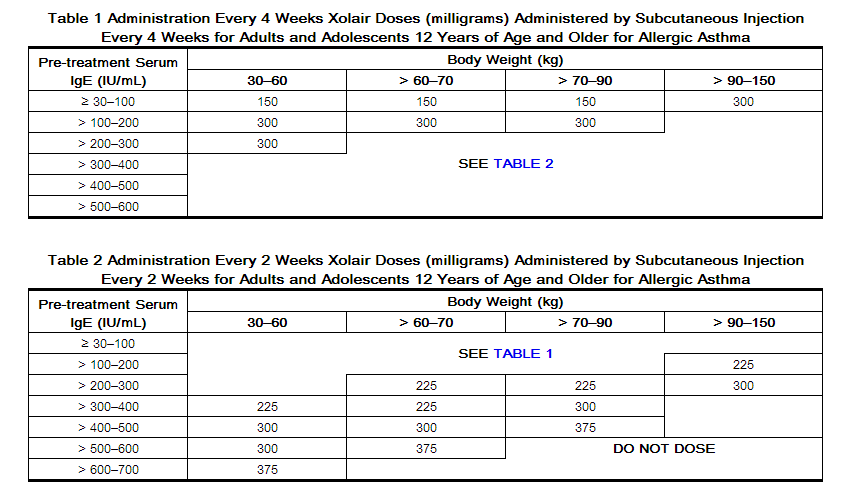
Revision of omalizumab dosing table for dosing every 4 instead of 2 weeks for specific ranges of bodyweight and baseline IgE - ScienceDirect

Revision of omalizumab dosing table for dosing every 4 instead of 2 weeks for specific ranges of bodyweight and baseline IgE - ScienceDirect

Revision of omalizumab dosing table for dosing every 4 instead of 2 weeks for specific ranges of bodyweight and baseline IgE - ScienceDirect